Role of Bcl-2 family proteins in apoptosis
We are investigating into the molecular mechanisms leading to the formation of pores in lipid membrane by the protein Bax, a protein of the Bcl-2 family.
What is apoptosis?
Apoptosis is a type of programmed cell death used by organisms to remove infected, abnormal, or otherwise undesired cells. It is a highly regulated process which involves many chemical signals and proteins. Despite, or because of this, it is possible for the process to malfunction, which may halt the onset of apoptosis (potentially resulting in cancer) or cause it to run uncontrolled (potentially resulting in organ failure or stroke).
How does a cell decide whether or not to die?
Many of the known apoptotic pathways involve the permabilization of the mitochondrial outer membrane, which then leaks several apogenic factors into the cell cytoplasm causing cell death. This is a regarded as the "decision point" in the overall apoptotic process, as once the outer mitochondrial membrane is permeabilized the cell is commited to dying. This step is regulated by the Bcl-2 family of proteins.
What are Bcl-2 family proteins?
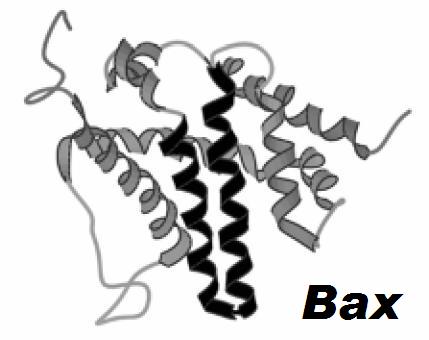
The Bcl-2 family of proteins is a group proteins sharing up to four sequence homology regions (BH1 to 4). Several proteins in this family also have structural homology although they are functionally very diverse. They can be pro-apoptotic (promoting mitochondrial outer membrane permeabilization) or anti-apoptotic (inhibiting mitochondrial outer membrane permeabilization).
Left: Solution structure of Bax. The α-helices involved in pore formation are outlined in black.
What are the questions addressed in our group?
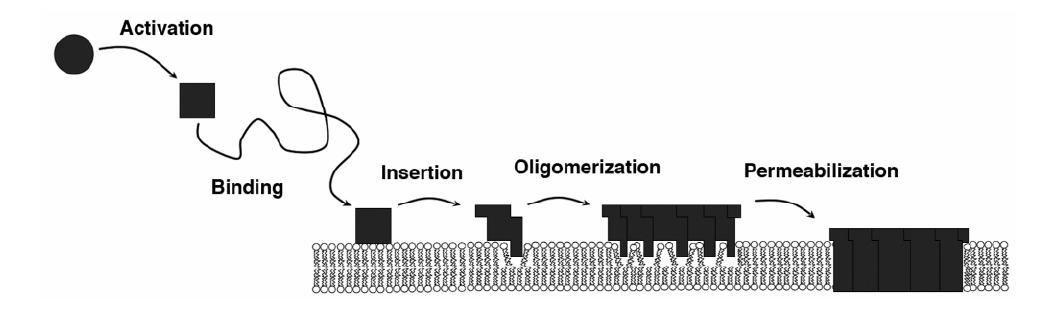
Our research revolves around the Bax protein, which has been shown to play a crucial role in apoptosis. Once activated, Bax joins the outer mitochondrial membrane and forms lipid pores, allowing for the diffusion of apogenic factors out of the mitochondria. Our group focuses on the molecular mechanisms of pore formation: structure and stoichiometry of the pores, conformation of the proteins in the membrane, influence of the lipids. We use a combination of fluorescence spectroscopy and scattering techniques that allow us to detail the conformation and movements of Bcl-2 family proteins in various environments.
Below: General mechanism for pore formation by pore-forming proteins (adopted from Fradin, Satsoura, Andrews 2009).
Movie
Publications
- Fradin, C., Satsoura, D., Andrews, D. W. (2009). Punching holes in membranes: How oligomeric pore-forming proteins and lipids cooperate to form aqueous channels in membranes. Handbook of Modern Biophysics, 2, 223-262. doi:10.1007/978-1-60761-314-5_9 [Read]
- Lovell, J. F., Billen, P. L., Bindner, S., Shamas-Din, A., Fradin, C., Leber, B., Andrews, D. W. (2008). Membrane binding by tBid initiates an ordered series of events culminating in membrane permeabilization by Bax. Cell, 135(6), 1074-1084. doi:10.1016/j.cell.2008.11.010 [Read]

